What Measures the Concentration and Strength of Hydrogen Peroxide
5 H 2 O 2 aq 2MnO. Remember that to prepare 1M of a substance you need the molar weight in 1L ie for 1M H2O2 youd need to dissolve 3401g of the substance in 1L.

Determination Of Hydrogen Peroxide Concentration
We wore gloves and lab coat as hydrogen peroxide is corrosive to the skin We wore goggles as hydrogen peroxide can cause serious eye problems.
. Measures the concentration and strength of hydrogen peroxide. CONCENTRATON 20 to 70 SCOPE. Use a sample size of about 03 g.
In this video I demonstrate a practical way to determine the concentration of a sample of hydrogen peroxide. Most patients who have inadvertently ingested 3 hydrogen peroxide can be observed safely at home if not symptomatic. It employs 4 ranges of calibration curves to enable the determination of.
DETERMINATION of HYDROGEN PEROXIDE. Researchers at Kanazawa University have successfully tested a sensor for measuring hydrogen peroxide concentrations near cell membranes. Hydrogen peroxide can be toxic if ingested inhaled or contacts the skin or eyes.
Determine the hydrogen peroxide content in at least three times and calculate. It is a special measure of concentration much like molarity or wv but only applicable to H02. The number 3 in this example and the sign or the word percent tell you this is the concentration.
Through reaction of the peroxide with ferrous iron monitored via a subsequent reaction with the dye xylenol orange and measurement of the absorbance of the solution at 550nm. If your hydrogen peroxide solution is 100 vol quite reasonable for a strong stock solution this means that 1000 cm3 of the H 2 O 2 produces 100000 cm3 of oxygen. The effects of hydrogen peroxide concentration on various physical properties such as weight loss absorbency GSM bursting strength and whiteness was studied to minimize the cost of bleaching process.
Biosensor measures extracellular hydrogen peroxide concentrations. Dye precursors that combine with hydrogen peroxide to form a larger permanent dye molecule. False - Volume measures the concentration and strength of hydrogen peroxide.
The photometric method is devised to measure the concentration of remaining permanganate after the reaction with residual hydrogen peroxide. Plastic syringes are used to measure and deliver a known volume of hydrogen peroxide solution and their nozzles should fit tightly into the hole in the flask bung. Determine the hydrogen peroxide in the sample of disinfectant.
Calculate by Hash Milhan is licensed under CC BY 20. Hydrogen Peroxide H2O2 is measured in wv which means number of grams in 100ml. If 417 moles of oxygen are produced then there must have been 417 x 2 moles of hydrogen peroxide 834 moles in 1 dm3 834 molar.
For example most hydrogen peroxide at the drugstore in brown bottles is 3 three percent. Mills can receive hydrogen peroxide at 70 concentration and dilute for storage at 50 concentration. The hair is fragile and may be close to breaking.
Natural vegetable and metallic hair colors. A concentration of 30 wv hydrogen peroxide implies that for every 100ml of the solution there is 30g of hygrogen peroxide. A Y x B.
1-Direct measurement of the absorbance at 240nm of the H2O2 molecule. If one has a sample of 10 volume strength H2O2 then each unit volume expressed in any unit. The primary effect is tissue injury due to oxidation of proteins.
This may trigger an OSHA requirement for a full Process Hazards Assessment. Acts as a stain to the hair. The classical methods for measuring hydrogen peroxide concentration are.
Patients who may have ingested a large amount of lower concentration solution or any amount of high concentration solution or who are symptomatic should be evaluated with. Sometimes concentration is also stated as PPM or parts per million. Hydrogen peroxide content determination.
Hydrogen peroxide measurement was based on the colorimetric method used to determine the millimolar quantities of hydrogen peroxide similarly to Graf. The classical methods for measuring hydrogen peroxide concentration are. The dilution calculator equation calculating in gallons is.
The reaction is oxidation-reduction and proceeds as shown below in net ionic form. Several processes in the human body are regulated by biochemical reactions. Use the same sample preparation and titration procedure as for the titer determination but dont heat up the sample solution.
The strength of the oxidizing reaction is determined by the concentration. Hydrogen peroxide solution H 2 O 2 aq IRRITANT at concentration used see CLEAPSS Hazcard HC050 and CLEAPSS Recipe Book RB045. In the titration method the residual hydrogen peroxide is estimated by titration with standard permanganate solution to estimate the hypochlorite concentration.
This method is designed for the determination of hydrogen peroxide in aqueous solutions containing 20 to 70 hydrogen peroxide. Their bursting strengths were measured by an Auto burst instrument following ISO 13938-1 method. The sensor has the potential to become a tool for new cancer therapies.
This is usually with water treatment or other very very. Hydrogen peroxide is an oxidizing agent. 30 is 30g in 100ml 12 is 12g in 100ml.
Viscosity measures the concentration and strength of hydrogen peroxide. Combustible material in the presence of high concentration hydrogen peroxide may quickly burst into flame. 1 volume strength H2O2 implies that one volume of it will liberate one volume of oxygen.
Concentration of hydrogen peroxide. The portion of hydrogen peroxide concentration range that you want to achieve in the final mixture. 3 hydrogen peroxide is a particular concentration.
Ignition with lower concentration hydrogen. 2-Through reaction of the peroxide with ferrous iron. Powdered off-the-scalp lighteners contain persulfate salts for quicker and stronger lightening.
Direct measurement of the absorbance at 240nm of the H2O2 molecule. Hydrogen peroxide in a diluted portion of the sample is quantitatively oxidized by titration with a potassium. Volume strength is a term unique to hydrogen peroxide.
Occasionally the release of oxygen may cause distension gastric or intestinal. Potentiometric Titration of Hydrogen Peroxide Buret One method of determining the concentration of a hydrogen peroxide H 2 O 2 solution is by titration with a solution of potassium permanganate KMnO 4 of known concentration. The volume of the final mix of purified water and hydrogen peroxide.

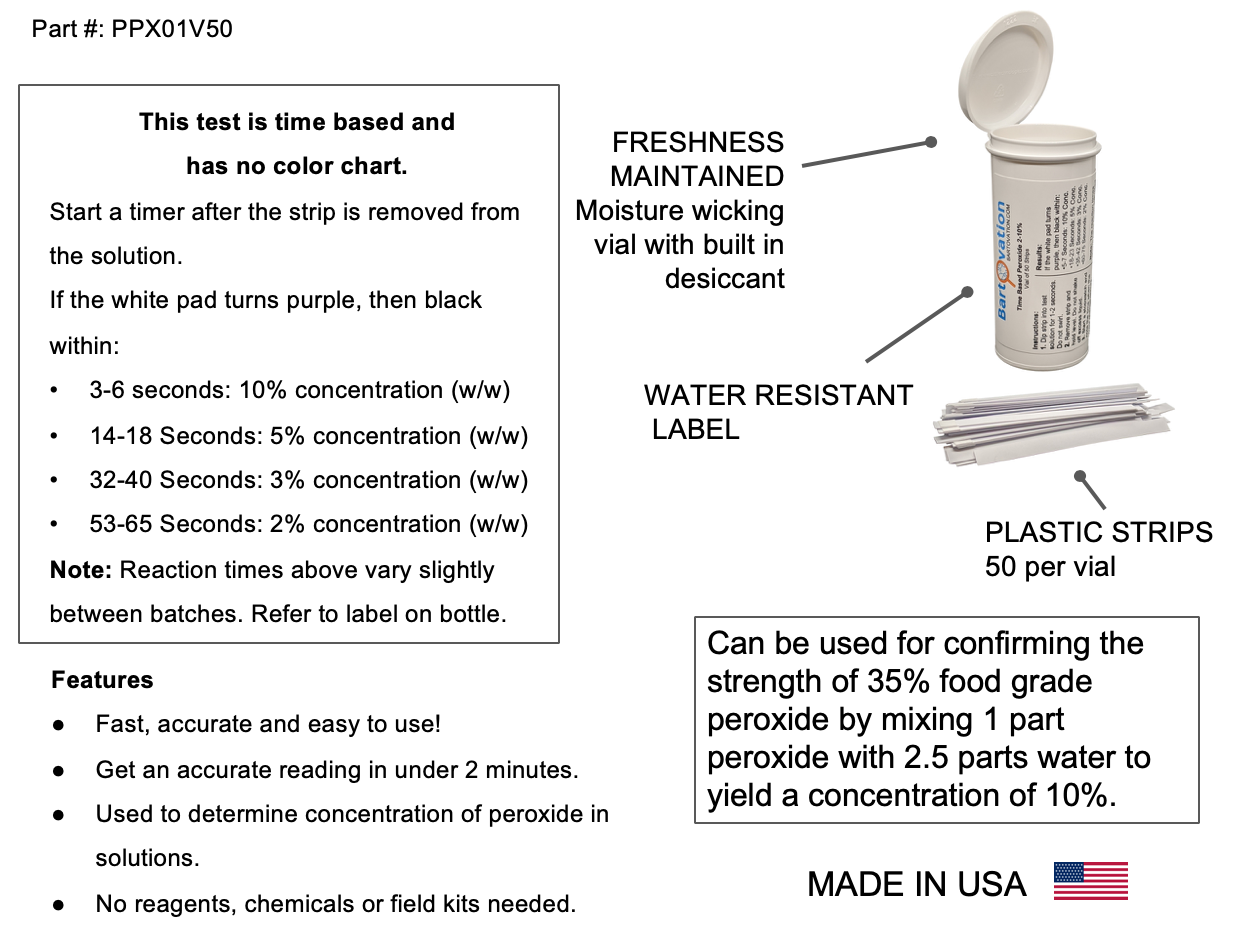
Very High Level Hydrogen Peroxide H2o2 Test Strips 2 10 Time Based Test Vial Of 50 Strips Bartovation Test Strips
Making High Concentrations Of Hydrogen Peroxide At Home Hackaday

Comments
Post a Comment